Oversolubility in the microvicinity of solid–solution interfaces
Abstract
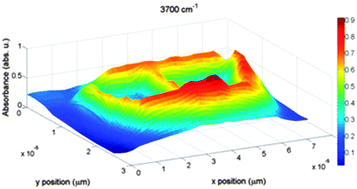
Water–solid interactions at the macroscopic level (beyond tens of nanometers) are often viewed as the coexistence of two bulk phases with a sharp interface in many areas spanning from biology to (geo)chemistry and various technological fields (membranes, microfluidics, coatings, etc.). Here we present experimental evidence indicating that such a view may be a significant oversimplification. High-resolution infrared and Raman experiments were performed in a 60 × 20 μm2 quartz cavity, synthetically created and initially filled with demineralized water. The IR mapping (3 × 3 μm2 beam size) performed using the SOLEIL synchrotron radiation source displays two important features: (i) the presence of a dangling free-OH component, a signature of hydrophobic inner walls; (ii) a shift of the OH-stretching band which essentially makes the 3200 cm−1 sub-band predominate over the usual main component at around 3400 cm−1. Raman maps confirmed these signatures (though less marked than IR's) and afforded a refined spatial distribution of this interfacial signal. This spatial resolution, statistically treated, results in a puzzling image of a 1–3 μm thick marked-liquid layer along the entire liquid–solid interface. The common view is then challenged by this strong evidence that a μm-thick layer analogous to an interphase forms at the solid–liquid interface. The thermodynamic counterpart of the vibrational shifts amounts to around +1 kJ mol−1 at the interface with a rapidly decreasing signature towards the cavity centre, meaning that vicinal water may form a reactive layer, of micrometer thickness, expected to have an elevated melting point, a depressed boiling temperature, and enhanced solvent properties.

 Please wait while we load your content...
Please wait while we load your content...