Beyond organic chemistry: aromaticity in atomic clusters
Abstract
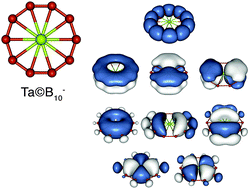
We describe joint experimental and theoretical studies carried out collaboratively in the authors' labs for understanding the structures and chemical bonding of novel atomic clusters, which exhibit aromaticity. The concept of aromaticity was first discovered to be useful in understanding the square-planar unit of Al4 in a series of MAl4− bimetallic clusters that led to discoveries of aromaticity in many metal cluster systems, including transition metals and similar cluster motifs in solid compounds. The concept of aromaticity has been found to be particularly powerful in understanding the stability and bonding in planar boron clusters, many of which have been shown to be analogous to polycyclic aromatic hydrocarbons in their π bonding. Stimulated by the multiple aromaticity in planar boron clusters, a design principle has been proposed for stable metal-cerntered aromatic molecular wheels of the general formula, M@Bnk−. A series of such borometallic aromatic wheel complexes have been produced in supersonic cluster beams and characterized experimentally and theoretically, including Ta@B10− and Nb@B10−, which exhibit the highest coordination number in two dimensions.
- This article is part of the themed collections: PCCP Perspectives and Electron delocalization and aromaticity: 150 years of the Kekulé benzene structure

 Please wait while we load your content...
Please wait while we load your content...