The nature of the silicaphilic fluorescence of PDMPO†
Abstract
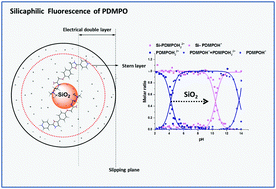
PDMPO (2-(4-pyridyl)-5-((4-(2-dimethylaminoethylaminocarbamoyl)methoxy)phenyl)oxazole), has unique silica specific fluorescence and is used in biology to understand biosilicification. This ‘silicaphilic’ fluorescence is not well understood nor is the response to local environmental variables like solvent and pH. We investigated PDMPO in a range of environments: using UV-vis and fluorescence spectroscopy supported by computational data, (SPARC, molecular dynamics simulations, density functional theory calculations), dynamic light scattering and zeta potential measurements to understand the PDMPO–silica interaction. From absorption data, PDMPO exhibited a pKa of 4.20 for PDMPOH22+ to PDMPOH+. Fluorescence emission measurements revealed large shifts in excited state pKa* values with different behaviour when bound to silica (pKa* of 10.4). PDMPO bound to silica particles is located in the Stern layer with the dye exhibiting pH dependent depolarising motion. In aqueous solution, PDMPO showed strong chromaticity with correlation between the maximum emission wavelength for PDMPOH+* and dielectric constant (4.8–80). Additional chromatic effects were attributed to changes in solvent accessible surface area. Chromatic effects were also observed for silica bound dye which allow its use as a direct probe of bulk pH over a range far in excess of what is possible for the dye alone (3–5.2). The unique combination of chromaticity and excited state dynamics allows PDMPO to monitor pH from 3 to 13 while also reporting on surface environment opening a new frontier in the quantitative understanding of (bio)silicification.

 Please wait while we load your content...
Please wait while we load your content...