Chemical bonding theory of single crystal growth and its application to crystal growth and design
Abstract
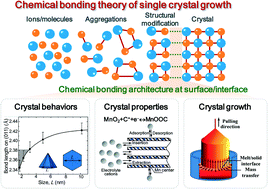
The effects of crystallization on the formation of geochemical, biological, and synthetic materials have been motivating decades of research into crystal nucleation and growth processes. The development of crystal growth theories and models can deepen the understanding of physicochemical interactions during the crystal growth process, which facilitates the designing of crystallization approaches in material production. The chemical bonding theory of single crystal growth emphasizes the dominant role of dynamic chemical bonding mechanisms at the growing interfaces. In this paper, we highlight the chemical bonding theory of single crystal growth from the chemical reaction viewpoint, by focusing on the atomic level of the growing interface between the liquid and crystal phases. Using ZnO, CeO2, MnO2 and Y3Al5O12 as examples, we review some typical applications of the chemical bonding theory of single crystal growth in calculating crystal habits, evaluating crystal properties, and guiding practical single crystal growth. Microscopically speaking, the essence of crystal growth and design is to create ideal chemical bonding architectures at both the crystal surface and the growing interface via both thermodynamic and kinetic strategies.

 Please wait while we load your content...
Please wait while we load your content...