Simultaneous determination of 14 components in Shuanghua Baihe tablets by ultrasonic-assisted extraction and HPLC-MS/MS
Abstract
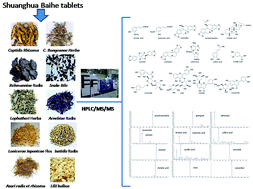
A simple, sensitive, selective, and reliable high-performance liquid chromatograph-tandem mass spectrometry (HPLC-MS/MS) method was developed and validated for the simultaneous detection and quantification of 14 compounds. These compounds were separated completely within 13 min on a Kinetex C18 core–shell column with acetonitrile and 0.1% formic acid as the mobile phase. The column was coupled to an ESI-triple-quadrupole mass spectrometer operated in positive and negative ion mode simultaneously. In total, 14 components were quantified in multiple reactions monitoring acquisition mode. The limits of detection of these compounds ranged from 0.12 to 2.10 ng mL−1, whereas the limits of quantitation ranged from 0.40 to 7.09 ng mL−1. All the calibration curves showed good linearities (r > 0.999) within the test ranges. An orthogonal design experiment was also used to investigate the effect of the extraction variables, including temperature, methanol to water ratio percent, and sonication time, on the ultrasound-assisted extraction (UAE). The optimum extraction condition and validated method was successfully used to analyze 10 batches of Shuanghua Baihe tablets (SBT) bought from Chinese markets. The method was validated to be simple, rapid, accurate and reliable to simultaneously determine 14 components in SBT and our results provide a new basis for the overall assessment of SBT quality.

 Please wait while we load your content...
Please wait while we load your content...