Complicity of haem in some adverse drug-reactions
Abstract
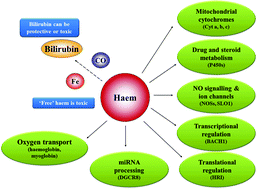
Adverse drug reactions are often idiosyncratic responses to administered medicines or herbal remedies that may be the consequence of multiple interactions between physiological and genetic factors with drug target and off-target effects. Identification and resolution of the mechanisms of these factors for individuals can prove problematic. Genetic variants or acquired fluctuations in the activity of the haem metabolism enzymes are well established predisposition factors in some known clinical hepatic disorders. Some of these disorders of nonerythroid haem supply can be triggered by drugs, hormones and other agents that normally have no untoward effects. Thus these mutations affecting the activity of the enzymes in the haem pathway can be regarded as predisposing genetic factors causing adverse reactions of some drugs in susceptible individuals. Carriers may be non-symptomatic for a prolonged period until the combination of factors such as consumption of medicinal products, alcohol, stress, hormone fluctuations and change in dietary habits trigger clinical symptoms. Despite these inherited or acquired changes of the haem metabolic pathway, many carriers do not develop symptoms illustrating the involvement of other unknown genetic components. In addition, haem is of fundamental cellular importance not only for respiratory cytochromes and oxygen transport but for many other roles such as for cytochrome P450, NO signalling, and microRNA formation. For these reasons, nonerythroid haem synthesis is under complex transcriptional and translational regulation linked to age, nutrition and circadian rhythm and to hormonal, drug and stress responses. Besides those already known, interference and genetic differences in the flux of supply, recruitment and use of haem for such fundamental processes may contribute to adverse drug reactions in metabolic arenas that are not yet recognised.
- This article is part of the themed collection: Celebrating the 2017 RSC Prize and Award Winners

 Please wait while we load your content...
Please wait while we load your content...