Gambogic acid suppresses cytochrome P450 3A4 by downregulating pregnane X receptor and up-regulating DEC1 in human hepatoma HepG2 cells
Abstract
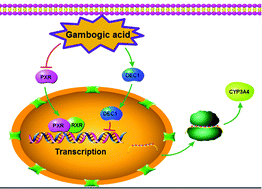
Gambogic acid (GA) was approved by the Chinese Food and Drug Administration for phase II clinical trials in patients with solid tumors. Recently, combinations of GA and chemotherapeutic agents have represented a promising therapeutic approach for cancer. However, little is known about the effect of GA on drug metabolism enzymes. In this study, we report for the first time that GA is a potent repressor of the cytochrome P450 3A4 (CYP3A4) enzyme, which is responsible for the oxidative metabolism of more than 60% of all pharmaceuticals. After HepG2 cells were treated with GA, the expression of CYP3A4 decreased at both the mRNA and protein levels, and CYP3A4 enzymatic activity was also reduced. Moreover, GA-mediated repression of CYP3A4 occurred after the decrease of PXR. Knockdown of PXR decreased CYP3A4 at the mRNA and protein levels, whereas PXR overexpression increased the CYP3A4 mRNA and protein levels. GA markedly decreased CYP3A4 expression in both normal and PXR overexpressing cells. Meanwhile, differentiated embryonic chondrocyte gene 1 (DEC1) expression rapidly increased in response to GA treatment, and subsequently, CYP3A4 expression continuously decreased. Overexpression of DEC1 decreased CYP3A4 mRNA and protein levels, and knockdown of DEC1 partially abolished the GA-mediated decrease in CYP3A4 expression. Taken together, our data suggest that two transcriptional factors, PXR and DEC1, are involved in the GA-mediated suppression of CYP3A4 in HepG2 cells.

 Please wait while we load your content...
Please wait while we load your content...