Cytotoxic effects and cellular oxidative mechanisms of metallic nanoparticles on renal tubular cells: impact of particle solubility
Abstract
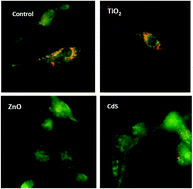
Many uncertainties remain regarding the potential toxic effect of nanoparticles (NPs). NPs can cross biological barriers, be carried in blood to kidneys, and damage renal cells. Yet, there is little data regarding NPs’ nephrotoxicity. The aim of this study was to understand the cytotoxic mechanisms induced by metallic NPs with different solubility properties (TiO2, ZnO, and CdS). Studies were performed in vitro on human epithelial tubular cells (HK-2). Cellular and molecular mechanisms were investigated through the oxidative stress status. Using the WST-1 assay it was found that the cytotoxicity of NPs was dependent on the particle size and metal solubility. Exposure to soluble CdS and ZnO NPs led to cell death in a dose-dependent manner related to release of metallic cations (Cd2+ and Zn2+). Insoluble TiO2 NPs had no cytotoxic effect. An analysis of ROS production and lipid peroxidation clearly revealed the involvement of oxidative stress in cell toxicity. Soluble ZnO and CdS NPs caused lysosomal membrane destabilization (acridine orange) and nuclear condensation (DAPI). A molecular approach was used for signaling pathways. ZnO and CdS NPs induced the translocation of Nrf2 and NF-κB and induction of antioxidant enzymes. TiO2 NPs did not cause lysosomal membrane destabilization or nuclear condensation. TiO2 NPs slightly activated Nrf2 nuclear translocation, but no significant NF-κB nuclear translocation was observed. For TiO2, the oxidative stress was not sufficient to trigger any membrane disruption. This study provides additional knowledge about the renal toxicity of NPs. The release of metal ions represents an important factor for determining the toxicity. This in vitro assay constitutes an additional step in nanomaterial safety assessment.
- This article is part of the themed collection: Nanotoxicology

 Please wait while we load your content...
Please wait while we load your content...