Bisphenol-A exposure alters memory consolidation and hippocampal CA1 spine formation through Wnt signaling in vivo and in vitro
Abstract
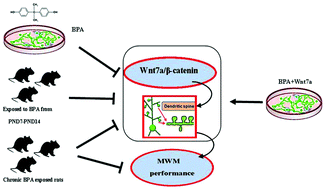
Bisphenol-A (BPA) has been implicated in the impairment of brain function, but the mechanism remains elusive. Our previous work has found that developmental BPA exposure impairs dendritic spine formation in the DG area of the hippocampus in rats. In the present study, we were to investigate the effects of BPA exposure on memory consolidation and its underlying mechanism, especially focusing on the canonical Wnt signaling pathway. BPA was administered to early developmental male Sprague-Dawley (SD) rat pups by intraperitoneal injection with BPA at the dosages of 50, 250 and 500 μg kg−1 per day for seven days (from postnatal day (PND) 7 to PND 14), as well as chronically exposed to male rats with dosages of 0.15 mg kg−1 per day and 7.50 mg kg−1 per day through drinking water containing BPA from the prenatal period to 12 weeks old. Further studies were conducted in cultured hippocampal neurons to observe the BPA-induced spine density changes and mechanism in vitro. The results showed that BPA exposure significantly impaired spatial memory in adult rats, accompanied by decreased hippocampal CA1 dendritic spine density. Besides, we observed dramatic changes of Wnt related proteins in BPA exposed little pups and cultured hippocampal neurons. In brief, β-catenin phosphorylation level was significantly increased and Wnt7a, one of its upstream ligands, was significantly decreased following BPA exposure. Additionally, in cultured hippocampal neurons, exogenous Wnt7a application reversed the BPA-induced dendritic spine impairment and the decreased β-catenin phosphorylation level. In summary, this study reported that BPA exposure may produce long-lasting effects and provide a novel mechanism for memory deficits induced by BPA.
- This article is part of the themed collection: New Talents

 Please wait while we load your content...
Please wait while we load your content...