Synthesis of novel nonlinear optical chromophores: achieving excellent electro-optic activity by introducing benzene derivative isolation groups into the bridge
Abstract
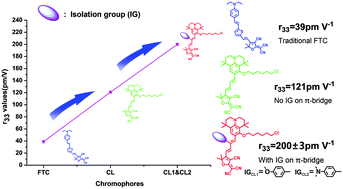
Three novel second order nonlinear optical (NLO) chromophores based on julolidinyl donors and tricyanofuran (TCF) acceptors linked together via modified polyene π-conjugation with rigid benzene derivative steric hindrance groups (chromophore CL1 and CL2) or unmodified polyene π-conjugation (chromophores CL) moieties as the bridges have been synthesized in good overall yields and systematically characterized. Density functional theory (DFT) was used to calculate the HOMO–LUMO energy gaps and first-order hyperpolarizability (β) of these chromophores. Besides, to determine the redox properties of these chromophores, cyclic voltammetry (CV) experiments were performed. Compared with CL, after introducing benzene derivative steric hindrance groups into the bridge, chromophores CL1 and CL2 had good thermal stabilities with high thermal decomposition temperatures which were 32 °C and 24 °C higher than chromophore CL, respectively. Most importantly, the introduction of rigid steric hindrance groups can effectively reduce dipole–dipole interactions to translate their relatively small β values into bulk high EO activities. By doping chromophores CL, CL1 and CL2 with a high loading of 45 wt% in APC, EO coefficients (r33) of up to 121, 197 and 202 pm V−1 at 1310 nm can be achieved, respectively. The r33 values of new chromophores CL1 and CL2 were about 1.6 times of chromophore CL. The high r33 value, good thermal stability and high yield suggest the potential use of the new chromophores in an nonlinear optical area.

 Please wait while we load your content...
Please wait while we load your content...