Tuning the oxidation potential of 2-phenylpyridine-based iridium complexes to improve the performance of bluish and white OLEDs†
Abstract
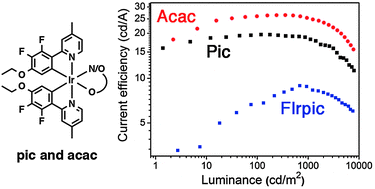
The use of electron-withdrawing substituents on the orthometalated phenyl ring is a common strategy to blue shift the emission of cyclometalated iridium complexes by stabilizing the highest occupied molecular orbital (HOMO), that is, increasing the oxidation potential of the complex. However, for application in blue organic light-emitting diodes (OLEDs), this approach imposes host materials with a deep HOMO, which negatively impacts the injection of charges, and hence the performance of the devices. In this context, we report new iridium complexes with an electron-donating substituent on the cyclometalated ligand to blue shift the emission while keeping a relatively low oxidation potential. As a result, bluish-green OLEDs based on host materials with shallow HOMOs (TCTA = 4,4′,4′′-tri(N-carbazolyl)-triphenylamine) display a higher performance than devices using FIrpic in the same architecture. The improvements are primarily attributed to the lower turn-on voltage (2.8 to 3 V) compared to those of FIrpic-device (3.6 V). White OLED was then prepared with a maximum brightness of 20 226 cd m−2 and current efficiency of 20.4 cd A−2 (at 100 cd m−2). Interestingly, a very small efficiency roll-off of about 1% at 1000 cd m−2 and high color stability were achieved. At a luminance level of 5000 cd m−2 the roll-off efficiency was still below 20%. The introduction of electron-donating substituents on a 2-phenylpyridine scaffold to obtain blue emitters with low oxidation potentials provides an alternative to strategies based on replacing the pyridine with imidazole, carbene, and pyrazole.
- This article is part of the themed collection: Small Molecules and Monodisperse Oligomers for Organic Electronics

 Please wait while we load your content...
Please wait while we load your content...