Resonance Raman spectroscopy and imaging of push–pull conjugated polymer–fullerene blends†
Abstract
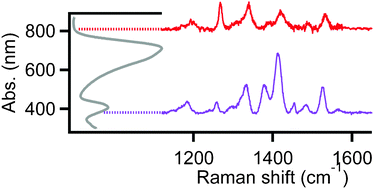
Blends of alternating ‘push–pull’ donor/acceptor (d/a) co-polymers with soluble fullerenes as active materials have shown promise for increasing power conversion efficiencies in organic photovoltaic (OPV) devices. We investigate morphology-dependent optical and electronic properties of poly[2,6-(4,4-bis-(2-ethylhexyl)-4H-cyclopenta[2,1-b;3,4-b′]dithiophene)-alt-4,7(2,1,3-benzothiadiazole)] (PCPDTBT) blended with [6,6]-phenyl C61 butyric acid methyl ester (PCBM) using electronic absorption and resonance Raman spectroscopies. Selective resonance excitation spanning the entire PCPDTBT absorption envelope (∼400–900 nm) was used to probe via Raman spectroscopy the degree of composition- and conformation-dependent charge transfer character along the polymer backbone. Raman intensities of characteristic PCPDTBT backbone donor/acceptor moieties vary with excitation wavelength. We perform density functional theory (DFT) calculations to assign Raman-active vibrational modes and correlate polymer backbone conformations to the degree of intra-chain donor/acceptor charge transfer character. We find the best agreement between experimental and simulated spectra for planarized PCPDTBT backbone consistent with strong charge transfer character along the backbone, which also gives rise to a new red-shifted absorption band appearing in PCBM blends. Resonance Raman and photocurrent imaging experiments were next used to spatially map morphology-dependent vibrational signatures of PCPDTBT donor/acceptor moieties within functioning solar cell devices. Solvent additives were applied using 1,8 octanedithiol (ODT) to modify PCPDTBT:PCBM morphologies and compared to as-cast blends. Raman and photocurrent images indicate a well-mixed morphology that we propose induces planarization of the PCPDTBT backbone.

 Please wait while we load your content...
Please wait while we load your content...