H2O-steered size/phase evolution and magnetic properties of large-scale, monodisperse FexOy nanomaterials†
Abstract
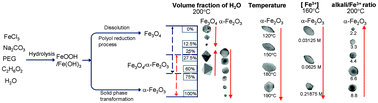
An easy mixed solvent solvothermal/hydrothermal method was developed for the one-step synthesis of monodisperse, single-crystal Fe3O4 and α-Fe2O3 nanomaterials. The morphologies can be varied from spherical, to octahedral, to rice like, and even to fusiform; the size can be continuously tuned to a range within 30–290 nm. The morphology-, dimension-, and phase-controlled growth of FexOy nanocrystals can be achieved by tuning kinetic factors, such as the H2O volume fraction (γ), Fe3+ concentration, reaction temperature, and the ratio of alkali/Fe3+. The threshold value γ (about 25%) for the H2O-steered size and phase evolutions was theoretically and experimentally inferred. The size- and phase-dependent saturation magnetization (Ms) and coercivity (Hc) were systematically investigated. High Ms was observed in single-crystal Fe3O4 nanomaterials because of high crystallinity; significantly enhanced Hc was exhibited by the as-obtained Fe3O4–α-Fe2O3 hybrid nanocrystals because of the unique unidirectional anisotropy and exchange bias. This study provided insights into the size and phase evolution mechanisms of nanocrystals in the EG–H2O system and served as efficient guidance for the tunable synthesis of monodisperse nanomaterials in a mixed solvent system. The uniform single-crystalline Fe3O4 and α-Fe2O3 nanomaterials can provide better platforms for studying their size- and phase-dependent optical, electric, and magnetic performances.

 Please wait while we load your content...
Please wait while we load your content...