Sequence-dependent mechanical, photophysical and electrical properties of pi-conjugated peptide hydrogelators†
Abstract
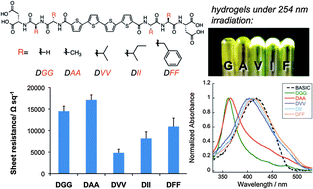
The ability to modulate intermolecular interactions in such a way as to impact nano-, micro- and even macroscale properties is an attractive aspect of self-assembling systems. We present an investigation of sequence-dependent rheological, photophysical and electrical properties of semiconducting peptide hydrogelators. Five different π-conjugated peptides containing a quaterthiophene core were studied, wherein the relative size and hydrophobicity of the amino acid residues adjacent to the π-electron core were varied in order to assess the impact of molecular variation on nanoscale and bulk material properties. Steady-state spectroscopic measurements of the peptides once assembled into 1D-nanostructures show distinct spectral characters as the relative size of the amino acid side chain adjacent to the π-electron core increases. Those peptides that formed hydrogels differed in network topography and rheological properties, with storage modulus (G′) values ranging from ∼3 to 20 kPa. The electrical properties of the peptide nanostructures were characterized by measuring the sheet resistance of dried peptide films on glass substrates. This study provides insights on the effects of amino acid sequence on the nanoscale to the macroscale electrical transport and mechanical properties of nanostructure-forming π-conjugated peptides.
- This article is part of the themed collection: Bioelectronics

 Please wait while we load your content...
Please wait while we load your content...