Bis-pyrene based reversibly multi-responsive fluorescence switches
Abstract
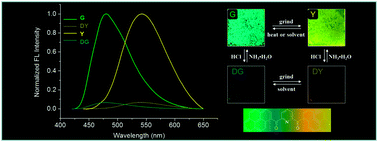
A bis-pyrene derivative m-Py-BP was designed and synthesized, where two pyrenes units were connected by pyridine-2,6-dicarbonyl linkers, displaying multi-responsive fluorescence switching behaviors. In solution, the quenched fluorescence emission of m-Py-BP by HCl was recovered with the addition of NH3·H2O. In the solid state, m-Py-BP exhibited vapor, mechano and thermo-induced fluorescence variations. The as-prepared sample showed green fluorescence (G form) with emission maxima at 483 nm. Upon mechanical shearing, the fluorescence of G form was transformed into a yellow color (Y form) by disrupting the crystalline structures. Vapor or heating induced the transformation from Y to G form. Powder X-ray diffraction (XRD), differential scanning calorimetry (DSC), time-resolved fluorescence (TRF) and scanning electron microscopy (SEM) supported the corresponding changes in supramolecular structures and morphologies. Interestingly, m-Py-BP showed acid–base responsive fluorescence switching behavior. The HCl could quench the fluorescence of Y form to DY (dark yellow) form, which could be recovered to Y form in the presence of NH3·H2O or converted to DG (dark green) form upon treatment with CHCl3 solvent vapor. Furthermore, the DG form could become DY form by mechanical shearing or convert to G form upon treatment with NH3·H2O vapor. The halochromic fluorescence should be related to the protonation of m-Py-BP molecules by acid treatment. The protonated pyridine became a strong electron acceptor, resulting in fluorescence quenching. m-Py-BP could construct a multi-responsive and recyclable fluorescence switching system.

 Please wait while we load your content...
Please wait while we load your content...