Silica-coated Gd(DOTA)-loaded protein nanoparticles enable magnetic resonance imaging of macrophages†
Abstract
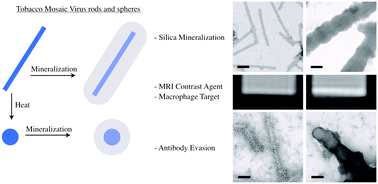
The molecular imaging of in vivo targets allows non-invasive disease diagnosis. Nanoparticles offer a promising platform for molecular imaging because they can deliver large payloads of imaging reagents to the site of disease. Magnetic resonance imaging (MRI) is often preferred for clinical diagnosis because it uses non-ionizing radiation and offers both high spatial resolution and excellent penetration. We have explored the use of plant viruses as the basis for MRI contrast reagents, specifically tobacco mosaic virus (TMV), which can assemble to form either stiff rods or spheres. We loaded TMV particles with paramagnetic Gd ions, increasing the ionic relaxivity compared to free Gd ions. The loaded TMV particles were then coated with silica maintaining high relaxivities. Interestingly, we found that when Gd(DOTA) was loaded into the interior channel of TMV and the exterior was coated with silica, the T1 relaxivities increased by three-fold from 10.9 mm−1 s−1 to 29.7 mm−1 s−1 at 60 MHz compared to uncoated Gd-loaded TMV. To test the performance of the contrast agents in a biological setting, we focused on interactions with macrophages because the active or passive targeting of immune cells is a popular strategy to investigate the cellular components involved in disease progression associated with inflammation. In vitro assays and phantom MRI experiments indicate efficient targeting and imaging of macrophages, an enhanced contrast-to-noise ratio was observed by shape-engineering (SNP > TMV) and silica-coating (Si-TMV/SNP > TMV/SNP). Because plant viruses are in the food chain, antibodies may be prevalent in the population. Therefore we investigated whether the silica-coating could prevent antibody recognition; indeed our data indicate that mineralization can be used as a stealth coating option to reduce clearance. Therefore, we conclude that the silica-coated protein-based contrast agent may provide an interesting candidate material for further investigation of in vivo delineation of disease through macrophage imaging.

 Please wait while we load your content...
Please wait while we load your content...