Oligoproline-derived nanocarrier for dual stimuli-responsive gene delivery†
Abstract
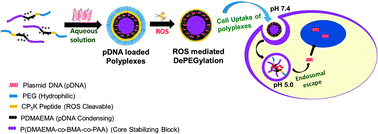
Gene therapy is a promising method for the treatment of vascular disease; however, successful strategies depend on the development of safe and effective delivery technologies with specific targeting to a diseased point of vasculature. Reactive oxygen species (ROS) are overproduced by vascular smooth muscle cells (VSMCs) at critical stages of atherosclerosis progression. Therefore, ROS were exploited as a stimulus for vascular targeted gene delivery in this study. A combination of bio-conjugation methods and controlled reverse addition-fragmentation chain-trasfer (RAFT) polymerization was utilized to synthesize a new ROS-cleavable, pH-responsive mPEG113-b-CP5K-b-PDMAEMA42-b-P(DMAEMA22-co-BMA40-co-PAA24) (PPDDBP) polymer as a nanocarrier for plasmid DNA (pDNA) delivery. The ros degradability of PPDDBP polymers was confirmed by SIN-1-mediated cleavage of CP5K peptide linkers through a shift in GPC chromatogram with an appearance of mPEG shoulder peak and an increase in zeta potential (ζ). The polyplex nanocarrier also demonstrated effective PDNA loading, serum stability, and hemocompatibility, indicating its excellent performance under physiological conditions. The polyplexes demonstrated ideal pH responsiveness for endosomal escape and effective ROS responsiveness for improved targeting in an in vitro model of pathogenic VSMCs in terms of both uptake and expression of reporter gene. These data suggest this novel nanocarrier polyplex system is a promising gene delivery tool for preventing or treating areas of high ROS, such as atherosclerotic lesions.

 Please wait while we load your content...
Please wait while we load your content...