Electrodeposition and pyrolysis of Mn/polypyrrole nanocomposites: a study based on soft X-ray absorption, fluorescence and photoelectron microspectroscopies
Abstract
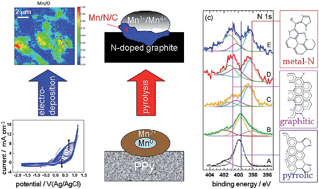
Electrodeposition of manganese/polypyrrole (Mn/PPy) nanocomposites has been recently shown to be a technologically relevant synthesis method for the fabrication of Oxygen Reduction Reaction (ORR) electrocatalysts. In this study we have grown such composites with a potentiostatic anodic/cathodic pulse-plating procedure and characterised them by a multi-technique approach, combining a suite of in situ and ex situ spectroscopic methods with electrochemical measurements. We have thus achieved a sound degree of molecular-level understanding of the hybrid co-electrodeposition process consisting of electropolymerisation of polypyrrole with incorporation of Mn. By in situ Raman spectroscopy we followed the formation of MnOx and the polymer by monitoring the build-up and development of the relevant vibrational bands. The compositional and chemical-state distribution of the as-deposited material has been investigated ex situ by soft X-ray fluorescence (XRF) mapping and micro-absorption spectroscopy (micro-XAS). XRF shows that the spatial distribution of Mn is consistent in a rather wide range of current densities (c.d.s), while micro-XAS reveals a mixture of Mn valencies, with higher oxidation states prevailing at higher c.d.s. Pyrolysis of electrodeposits, desirable for obtaining more durable and active catalysts, has been followed in situ by photoelectron microspectroscopy, allowing to assess the evolution of: (i) the electrodeposit morphology, resulting in a uniform distribution of nanoparticles; (ii) the chemical state of manganese, changing from a mixture of valences to a final state consisting of Mn(III) and Mn(IV) oxides and (iii) the bonding nature of nitrogen, from initially N-pyrrolic to a combination of pyridinic and Mn–N/graphitic.


 Please wait while we load your content...
Please wait while we load your content...