Stability of NdBaCo2−xMnxO5+δ (x = 0, 0.5) layered perovskites under humid conditions investigated by high-temperature in situ neutron powder diffraction†
Abstract
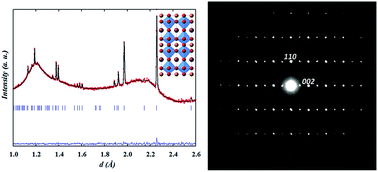
The double perovskites NdBaCo2−xMnxO5+δ (x = 0 and 0.5) were investigated using in situ high temperature neutron powder diffraction in dry argon and wet atmospheres (40% D2O/argon and 40% D2O/air) in order to assess their stability as cathodes in proton conducting fuel cells. The x = 0 oxide loses oxygen on heating in dry argon at T > 400 °C and exhibits an oxygen vacancy order–disorder transition as evidenced by the orthorhombic Pmmm to tetragonal P4/mmm transition. Refinement of site occupancy factors suggests that the oxygen vacancies mainly form in the Nd layers and to a lesser extent at the equatorial positions of the transition metal polyhedra; at 800 °C, δ ∼ 0. When the gas was changed to wet argon at 800 °C and the sample cooled to 260 °C, no structural modification or change in the oxygen content was detected and no impurity phases formed, highlighting the excellent stability of the sample in wet atmospheres. On switching the gas to wet air at 260 °C, thermal analysis and neutron powder diffraction data together reveal that the sample intercalates mainly oxygen rather than proton defects within a two-phase process involving two orthorhombic phases, reflecting the symmetry of the reduced and oxidised materials. On heating, the sample transforms at T ≥ 600 °C to a single tetragonal phase whose symmetry is retained up to 800 °C and on subsequent cooling. The x = 0.5 material prepared in argon adopted a tetragonal P4/mmm structure at RT with δ ∼ 0. Its symmetry remains tetragonal on heating/cooling in wet argon. On changing the gas to wet air at 260 °C, it takes up oxygen via a two-phase process involving two tetragonal phases. Since fast oxidation is the main process that fills the oxygen vacancies of these double perovskites in wet air, a large oxygen deficiency seems to be not the only requirement for effective proton incorporation in this family of materials with basic characteristics.

 Please wait while we load your content...
Please wait while we load your content...