An electrochemical sensing platform based on a reduced graphene oxide–cobalt oxide nanocube@platinum nanocomposite for nitric oxide detection†
Abstract
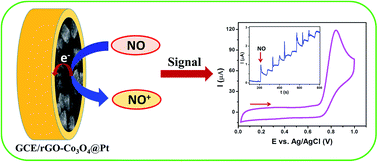
We report a facile one-pot hydrothermal synthesis of a reduced graphene oxide–cobalt oxide nanocube@platinum (rGO–Co3O4@Pt) nanocomposite and its application toward the electrochemical detection of nitric oxide (NO). The rGO–Co3O4@Pt nanocomposite was characterized by field emission scanning electron microscopy (FESEM), energy dispersive X-ray (EDX) mapping, X-ray diffraction (XRD) and Raman analyses. The nanocomposite modified glassy carbon (GC) electrode was used for the electrochemical oxidation of nitric oxide (NO) and it showed better catalytic performance in terms of catalytic peak current and shift in overpotential when compared to those of rGO, Co3O4 nanocubes and rGO–Co3O4 nanocomposite modified electrodes. The rGO–Co3O4@Pt nanocomposite modified electrode showed a better sensing ability toward the in situ generated NO in NO2− containing phosphate buffer solution (PBS) than the other controlled modified electrodes. The Pt nanoparticles present in the nanocomposite could enhance the sensing performance and the limit of detection (LOD) was found to be 1.73 μM with a signal-to-noise (S/N) ratio of ∼3 using the amperometric i–t curve technique. Furthermore, the nanocomposite modified electrode showed selectivity toward the detection of NO in the presence of a 100-fold higher concentration of other physiologically important analytes. The proposed sensor was stable, reproducible and selective toward the detection of NO.


 Please wait while we load your content...
Please wait while we load your content...