Reaction mechanism from quantum molecular dynamics for the initial thermal decomposition of 2,4,6-triamino-1,3,5-triazine-1,3,5-trioxide (MTO) and 2,4,6-trinitro-1,3,5-triazine-1,3,5-trioxide (MTO3N), promising green energetic materials†
Abstract
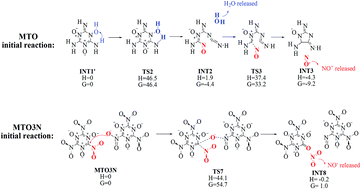
Klapötke and co-workers recently designed two new materials, 2,4,6-triamino-1,3,5-triazine-1,3,5-trioxide (MTO) and 2,4,6-trinitro-1,3,5-triazine-1,3,5-trioxide (MTO3N), envisioned as candidates for green high-energy materials. However, all attempts at synthesis have failed. In order to validate the expected properties for these systems and to determine why these materials are too unstable to synthesize, we used the PBE flavor of Density Functional Theory (DFT) to predict the crystal structures for MTO and MTO3N and then we carried out DFT molecular dynamics simulations (DFT-MD) to determine the initial reaction mechanisms for decomposition. Klapötke estimated that MTO would have a density of ρ = 1.859 g cm−3 with an estimated detonation velocity (Dv) of 8.979 km s−1, making it comparable to RDX (ρ = 1.82 g cm−3, Dv = 8.75 km s−1) and β-HMX (ρ = 1.91 g cm−3, Dv = 9.10 km s−1). His estimated impact sensitivity >30 J, make it much better than HMX (7 J) and RDX (7.5 J). Our predicted crystal structure for MTO (P2(1) space group) leads to ρ = 1.859 g cm−3, in good agreement with expectations. Our DFT-MD studies find that the first step in the decomposition of MTO is intermolecular hydrogen-transfer reaction (barrier 3.0 kcal mol−1) which is followed quickly by H2O and NO release with reaction barriers of 46.5 and 35.5 kcal mol−1. In contrast for MTO3N (P2(1)/c predicted space group), we find that the first steps are a bimolecular decomposition to release NO2 (ΔH = 44.1 kcal mol−1, ΔG = 54.7 kcal mol−1) simultaneous with unimolecular NO2 cleavage (ΔH = 59.9 and ΔG = 58.2 kcal mol−1) a unique initial reaction among EMs. These results suggest that MTO3N would be significantly more thermally stabile (barrier > 6.0 kcal mol−1 higher) than RDX and HMX, making it an excellent candidate to be insensitive new green energetic materials. However we find that MTO leads to very favorable hydrogen transfer reactions that may complicate synthesis and crystallization, making MTO3N the more promising system.

 Please wait while we load your content...
Please wait while we load your content...