Improved oxide-ion conductivity of NdBaInO4 by Sr doping†
Abstract
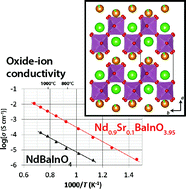
The oxide-ion conductivity of NdBaInO4 has been increased by Sr doping. Nd0.9Sr0.1BaInO3.95 showed the highest electrical conductivity among Nd1−xSrxBaInO4−x/2 (x = 0.0, 0.1, 0.2, and 0.3). The oxide-ion conductivity σion of Nd0.9Sr0.1BaInO3.95 (σion = 7.7 × 10−4 S cm−1) is about 20 times higher than that of NdBaInO4 (σion = 3.6 × 10−5 S cm−1) at 858 °C, and the activation energy of oxide-ion conduction is a little lower for Nd0.9Sr0.1BaInO3.95 (0.795(10) eV) than that for NdBaInO4 (0.91(4) eV). The structure analysis based on neutron powder diffraction data revealed that the Sr exists at the Nd site and oxygen vacancies are observed in Nd0.9Sr0.1BaInO3.95. This result indicates that the increase of the oxide-ion conductivity is mainly due to the increase of the carrier concentration. The bond valence-based energy landscape indicated two-dimensional oxide-ion diffusion in the (Nd,Sr)2O3 unit on the bc-plane and a decrease of the energy barrier by the substitution of Nd with Sr cations.

 Please wait while we load your content...
Please wait while we load your content...