Solar thermochemical conversion of CO2 into fuel via two-step redox cycling of non-stoichiometric Mn-containing perovskite oxides†
Abstract
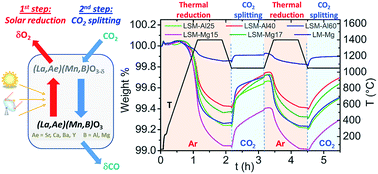
Solar-driven two-step thermochemical splitting of carbon dioxide represents one of the most promising candidate processes for carbon-neutral fuel production and solar energy storage. This redox process yields CO, one of the main precursors for liquid hydrocarbon fuels, and inherently results in the recycling and valorization of the CO2 greenhouse gas. In this context, non-stoichiometric perovskites such as La1−xSrxMnO3−δ have emerged as attractive candidate redox materials because of their large oxygen storage capacities and exchange capabilities that are required for applications in high temperature thermochemical redox cycles for splitting CO2. This study addresses the investigation of reactive Mn-containing substituted perovskites derived from this La1−xSrxMnO3−δ series, where Ba2+, Ca2+ or Y3+ have been incorporated on the A-site. Conversely, introduction of Al3+ and Mg2+ on the B-site was also examined. The materials were synthesized and their structures were validated by powder X-ray diffraction. Their thermochemical redox performances were subsequently quantified by thermogravimetric analysis, while the microstructure of the polycrystalline materials was examined by scanning electron microscopy. High redox activity for CO2 splitting was observed for the majority of the substituted perovskites examined here, in which Mn was the single reducible cation and Mn4+/Mn3+ redox pair was activated in the oxygen-exchange process. The structural evolutions and thermochemical behavior are discussed with respect to the distinct chemistries of the additional cations incorporated in these Mn-based perovskites.

 Please wait while we load your content...
Please wait while we load your content...