Highly selective CO2 photoreduction to CO over g-C3N4/Bi2WO6 composites under visible light†
Abstract
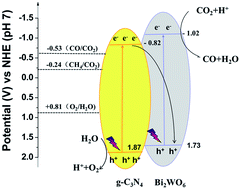
CO2 is highly stable and therefore extremely difficult to be reduced at room temperature even by photocatalysis. Herein, a series of g-C3N4/Bi2WO6 composites have been synthesized through a facile in situ hydrothermal approach, which demonstrated greatly enhanced response to visible light, and consequently a remarkably enhanced CO2 selective photoreduction to CO. The g-C3N4 content and synthesis parameters of these composites have been tuned to obtain the optimized photocatalytic activity with a peak CO production rate of 5.19 μmol g−1 h−1 under visible light irradiation at room temperature, which was 22 and 6.4 times that on pure g-C3N4 and Bi2WO6, respectively. Based on the matched band energy potentials between g-C3N4 and Bi2WO6 in the synthesized composites, a possible Z-scheme mechanism, which features a significantly promoted separation of photo-generated carriers under visible light irradiation by the composites, has been proposed to account for the distinctive CO2 photoreduction performance.

 Please wait while we load your content...
Please wait while we load your content...