Synthesis of a nitrogen rich (2D–1D) hybrid carbon nanomaterial using a MnO2 nanorod template for high performance Li-ion battery applications†
Abstract
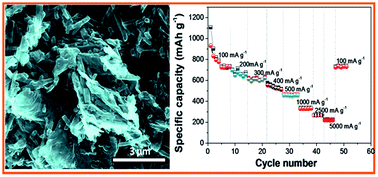
A novel strategy is developed to synthesize a nitrogen rich few layer graphene–carbon nanotube [N-(FLG–CN)] hybrid material with the help of self-degradable MnO2 nanorod templates for the application as the anode electrode with high performance and long-term cyclic stability in Li ion batteries. During the synthesis procedure, the surfaces of MnO2 nanorods and few layer graphene (FLG) are non-covalently functionalized with anionic and cationic polyelectrolytes, respectively, for proper mixing of the constituents. Polymerization of this one- and two-dimensional hybrid composite with a nitrogen containing polymer and subsequent pyrolysis at 800 °C temperature lead to the formation of a highly porous nitrogen doped-(FLG–CN) hybrid nanocomposite with a nitrogen doping level of 9.3 wt%. The N-(FLG–CN) electrode material in Li ion batteries displays superior reversible capacities of 739 mA h g−1 after the 30th cycle at a current density of 100 mA g−1 and 445 mA h g−1 after the 500th cycle at a high current density of 500 mA g−1. As compared to the pristine graphene material, the N doped (FLG–CN) hybrid material shows a two-fold enhancement in specific capacity at high current densities (∼5000 mA g−1) and long-term cyclic stability (1000 cycles). The highly defective and porous 1D–2D morphology of the N-(FLG–CN) hybrid structure gives more adsorption sites for lithium ions, meanwhile nitrogen doping significantly reduces the charge transfer resistance of graphene based electrodes.

 Please wait while we load your content...
Please wait while we load your content...