Spontaneous grafting of 9,10-phenanthrenequinone on porous carbon as an active electrode material in an electrochemical capacitor in an alkaline electrolyte†
Abstract
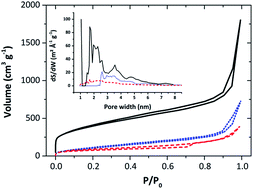
Spontaneous grafting of 9,10-phenanthrenequinone (PQ) on Black Pearls carbon by reduction of the corresponding in situ generated diazonium cations has been successfully achieved. Black Pearls was also modified by adsorption from a PQ/acetonitrile solution. Nitrogen gas adsorption measurements revealed that the chemisorption and adsorption of PQ molecules caused a significant decrease of the BET surface area and affected mainly the microporosity of the carbon powder. A detailed characterization by transmission electron microscopy, thermogravimetric analysis, X-ray photoelectron spectroscopy, and electrochemical techniques confirmed the immobilization of PQ molecules at the surface and suggested the formation of a covalent bond between the molecule and the substrate during spontaneous grafting. The loading of PQ molecules on Black Pearls was controlled by changing the concentration of the amine precursor. The superior capacity retention of PQ-grafted Black Pearls electrodes relative to PQ-adsorbed Black Pearls electrodes upon cycling in an alkaline electrolyte provides indirect evidence for the covalent linkage of PQ to the carbon substrate. Finally, a slight modification of the quinone structure leads to a significant difference of the stability of the modified electrodes. PQ-grafted Black Pearls electrodes were found to be much more stable than anthraquinone-grafted Black Pearls electrodes suggesting a better grafting efficiency. Moreover, the proximity of the ketone functionalities in PQ could stabilize the molecule at the carbon surface.

 Please wait while we load your content...
Please wait while we load your content...