Shape control of lipid bilayer membranes by confined actin bundles†
Abstract
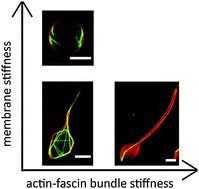
In living cells, lipid membranes and biopolymers determine each other's conformation in a delicate force balance. Cellular polymers such as actin filaments are strongly confined by the plasma membrane in cell protrusions such as lamellipodia and filopodia. Conversely, protrusion formation is facilitated by actin-driven membrane deformation and these protrusions are maintained by dense actin networks or bundles of actin filaments. Here we investigate the mechanical interplay between actin bundles and lipid bilayer membranes by reconstituting a minimal model system based on cell-sized liposomes with encapsulated actin filaments bundled by fascin. To address the competition between the deformability of the membrane and the enclosed actin bundles, we tune the bundle stiffness (through the fascin-to-actin molar ratio) and the membrane rigidity (through protein decoration). Using confocal microscopy and quantitative image analysis, we show that actin bundles deform the liposomes into a rich set of morphologies. For liposomes having a small membrane bending rigidity, the actin bundles tend to generate finger-like membrane protrusions that resemble cellular filopodia. Stiffer bundles formed at high crosslink density stay straight in the liposome body, whereas softer bundles formed at low crosslink density are bent and kinked. When the membrane has a large bending rigidity, membrane protrusions are suppressed. In this case, membrane enclosure forces the actin bundles to organize into cortical rings, to minimize the energy cost associated with filament bending. Our results highlight the importance of taking into account mechanical interactions between the actin cytoskeleton and the membrane to understand cell shape control.

 Please wait while we load your content...
Please wait while we load your content...