Abstract
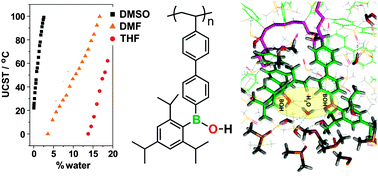
The thermo-responsive properties of borinic acid polymers were investigated by experimental and molecular dynamics simulation studies. The homopolymer poly(styrylphenyl(tri-iso-propylphenyl)borinic acid) (PBA) exhibits an upper critical solution temperature (UCST) in polar organic solvents that is tunable over a wide temperature range by addition of small amounts of H2O. The UCST of a 1 mg mL−1 PBA solution in DMSO can be adjusted from 20 to 100 °C by varying the H2O content from ∼0–2.5%, in DMF from 0 to 100 °C (∼3–17% H2O content), and in THF from 0 to 60 °C (∼4–19% H2O). The UCST increases almost linearly from the freezing point of the solvent with higher freezing point to the boiling point of the solvent with the lower boiling point. The mechanistic aspects of this process were investigated by molecular dynamics simulations. The latter indicate rapid and strong hydrogen-bond formation between BOH moieties and H2O molecules, which serve as crosslinkers to form an insoluble network. Our results suggest that borinic acid-containing polymers are promising as new “smart” materials, which display thermo-responsive properties that are tunable over a wide temperature range.

 Please wait while we load your content...
Please wait while we load your content...