Synthesis and high-throughput characterization of structural analogues of molecular glassformers: 1,3,5-trisarylbenzenes†
Abstract
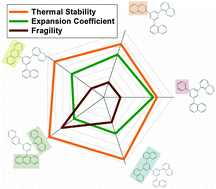
We report the synthesis and characterization of an analogous series of small organic molecules derived from a well-known glass former, 1,3-bis(1-naphthyl)-5-(2-naphthyl)benzene (α,α,β-TNB). Synthesized molecules include α,α,β-TNB, 3,5-di(naphthalen-1-yl)-1-phenylbenzene (α,α-P), 9-(3,5-di(naphthalen-1-yl)phenyl)anthracene (α,α-A), 9,9′-(5-(naphthalen-2-yl)-1,3-phenylene)dianthracene (β-AA) and 3,3′,5,5′-tetra(naphthalen-1-yl)-1,1′-biphenyl (α,α,α,α-TNBP). The design of molecules was based on increasing molecular weight with varied π–π interactions in one or more substituents. The synthesis is based on Suzuki cross-coupling of 1-bromo-3-chloro-5-iodobenzene with arylboronic acids, which allows attachment of various substituents to tailor the chemical structure. The bulk compounds were characterized using NMR spectroscopy and differential scanning calorimetry (DSC). Thin films of these compounds were produced using physical vapor deposition and were subsequently annealed above the glass transition temperatures (Tg). For each molecular glass, cooling rate-dependent glass transition temperature measurements (CR-Tg) were performed using ellipsometry as a high-throughput method to characterize thin film properties. CR-Tg allows rapid characterization of glassy properties, such as Tg, apparent thermal expansion coefficients, apparent activation energy at Tg and fragility. DSC measurements confirmed the general trend that increasing molecular weight leads to increasing melting point (Tm) and Tg. Furthermore, CR-Tg provided evidence that the introduction of stronger π-interacting substituents in the chosen set of structural analogues increases fragility and decreases the ability to form glasses, such that β-AA has the largest fragility and highest tendency to crystallize among all the compounds. These strong interactions also significantly elevate Tg and promote more harmonic intermolecular potentials, as observed by decreasing value of the apparent thermal expansion coefficient.

 Please wait while we load your content...
Please wait while we load your content...