A quantitative definition of hypervalency†
Abstract
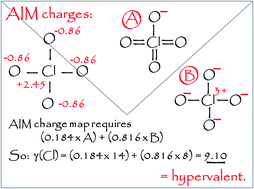
From the inception of Lewis' theory of chemical bonding, hypervalency has remained a point of difficulty that has not been fully resolved by the currently accepted qualitative definition of this term. Therefore, in this work, a quantitative measure of hypervalency has been developed. The only required input is the atomic charge map, which can be obtained from either quantum calculations or from experiment. Using this definition, it is found that well-known species such as O3, CH2N2 and ClO4−, are indeed hypervalent, whilst others such as XeF4, PCl5 and SO42−, are not. Quantitative analysis of known species of general formulae XFnm−, XClnm−, and XOnm− shows that there are no fundamental differences in chemical bonding for hypervalent and non-hypervalent species. Nevertheless, hypervalency is associated with chemical instability, as well as a high degree of covalent rather than ionic bonding. The implications for accepted Lewis structure conventions are discussed.
- This article is part of the themed collection: ISACS17: Challenges in Chemical Renewable Energy


 Please wait while we load your content...
Please wait while we load your content...