Cyclometalated iridium(iii) complexes as lysosome-targeted photodynamic anticancer and real-time tracking agents†
Abstract
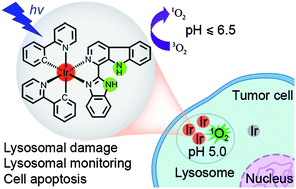
Stimuli-activatable photosensitizers (PSs) are highly desirable for photodynamic therapy (PDT) to selectively demolish tumor cells. On the other hand, lysosomes are emerging as attractive anticancer targets. Herein, four cyclometalated iridium(III)–β-carboline complexes with pH-responsive singlet oxygen (1O2) production and lysosome-specific imaging properties have been designed and synthesized. Upon visible light (425 nm) irradiation, they show highly selective phototoxicities against cancer cells. Notably, complex 2 ([Ir(N^C)2(N^N)](PF6) in which N^C = 2-phenylpyridine and N^N = 1-(2-benzimidazolyl)-β-carboline) displays a remarkably high phototoxicity index (PI = IC50 in the dark/IC50 in light) of >833 against human lung carcinoma A549 cells. Further studies show that 2-mediated PDT induces caspase-dependent apoptosis through lysosomal damage. The pH-responsive phosphorescence of complex 2 can be utilized to monitor the lysosomal integrity upon PDT, which provides a reliable and convenient method for in situ monitoring of therapeutic effect and real-time assessment of treatment outcome. Our work provides a strategy for the construction of highly effective multifunctional subcellular targeted photodynamic anticancer agents through rational structural modification of phosphorescent metal complexes.
- This article is part of the themed collection: Global challenges: Health & Food

 Please wait while we load your content...
Please wait while we load your content...