Dynamic induction of enantiomeric excess from a prochiral azobenzene dimer under circularly polarized light†
Abstract
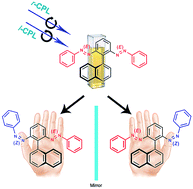
The ability to photoinduce enantiomeric excess from the chirality of circularly polarized light (CPL) is pertinent to the study of the origin of homochirality in biomolecules. Such CPL-induced reactions, including both chirality generation and formation of partial enantiomeric imbalance, from nonchiral starting compounds have been known, however, only for the conversion of diarylolefins into chiral helicenes. In this study we synthesized three different prochiral molecules, each featuring a pair of photoisomerizable phenylazo moieties arranged symmetrically upon the phenyl rings of an sp3-hybridized carbon atom (1), the phenyl rings of [2.2]paracyclophane (2), and the ortho positions of a phenyl ring bearing a naphthyl unit (3), and then investigated the possibility of photoinducing enantiomeric excess under CPL. Irradiation of 1–3 with light induced E ↔ Z photoisomerizations of their azobenzene moieties, giving mixtures of their EE, EZ, and ZZ isomers in the photostationary state (PSS). Among these regioisomers, the EZ forms are chiral and existed as racemic mixtures of R and S stereoisomers. Upon CPL irradiation of 3, circular dichroism (CD) revealed enantiomeric enrichment of one of the EZ stereoisomers; furthermore, irradiation with r- or l-CPL gave CD signals opposite in sign, but with equal intensity, in the PSS. In contrast, 1 and 2 did not give any detectable induced CD upon CPL irradiation. These experimental results can be explained by considering the different Kuhn anisotropy factors (g) of the (R)-EZ and (S)-EZ stereoisomers of 1–3, assuming that the origin of the enantiomeric excess is the enantio-differentiating photoisomerization from EZ stereoisomers to nonchiral EE or ZZ regioisomers by r- or l-CPL. In short, we demonstrate the simultaneous induction of chirality and enantiomeric excess from a prochiral azobenzene dimer via a chiral regioisomer formed in situ upon CPL irradiation.

 Please wait while we load your content...
Please wait while we load your content...