Synthesis and unimolecular micelles of amphiphilic copolymer with dendritic poly(l-lactide) core and poly(ethylene oxide) shell for drug delivery†
Abstract
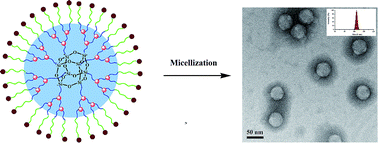
A novel type of amphiphilic copolymer POSS-(G3-PLLA-b-PEO-COOH)8 with a hydrophobic third-generation dendritic PLLA core and a functionalized hydrophilic PEO shell with surface carboxylic groups was synthesized as a carrier for drug delivery. The POSS-(G3-PLLA-OH)8 core was first synthesized by the combination of repetitive ring-opening polymerization (ROP) of L-lactide and branching reactions. Second, the amphiphilic copolymer POSS-(G3-PLLA-b-PEO-COOH)8 was obtained by esterification coupling between allyl-PEO-COOH chains and POSS-(G3-PLLA-OH)8, followed by the peripheral allyl groups reacting with 3-mercaptopropionic acid (MPA). In aqueous solution, this amphiphilic copolymer exists as stable unimolecular micelles with a unique core–shell structure and uniform size distribution (99.9–102.5 nm), as detected by dynamic light scattering (DLS) and transmission electron microscopy (TEM). Doxorubicin (DOX), an anticancer drug, was encapsulated into POSS-(G3-PLLA-b-PEO-COOH)8 micelles to evaluate the drug release profile. The result showed that the DOX-loaded micelles, with a loading content 18.5 ± 2.3 w/w%, exhibited controlled release of up to 39% loaded drug over a time period of 80 h. In addition, the surface carboxylic groups provide the opportunity for further conjugating targeting molecules, fluorescence dye or even drugs. These results indicated that the structurally stable unimolecular micelles from POSS-(G3-PLLA-b-PEO-COOH)8 hold potential applications as controlled drug delivery nanocarriers.

 Please wait while we load your content...
Please wait while we load your content...