Lithium iron phosphate (LiFePO4) as an effective activator for degradation of organic dyes in water in the presence of persulfate
Abstract
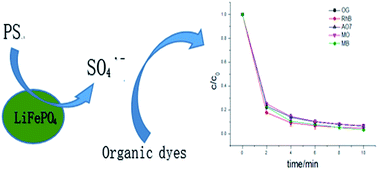
Lithium iron phosphate (LiFePO4) was prepared and applied in the heterogeneous activation of persulfate (PS) for Orange G (OG) degradation in water. Scanning electron microscopy, X-ray diffraction, Raman spectroscopy and X-ray photoelectron spectroscopy (XPS) were used to characterize the properties of the material before and after activation. The effects of several parameters on its catalytic activity were also investigated. It was found that the catalyst presented ultrafast activity for OG and other organic dye degradations. The results from XPS further suggested that the highly catalytic efficiency possibly involved the activation of PS to sulfate radicals as main radical meditated by the redox pair of Fe(II)/Fe(III) of LiFePO4/FePO4.

 Please wait while we load your content...
Please wait while we load your content...