Kaempferide, the most active among the four flavonoids isolated and characterized from Chromolaena odorata, induces apoptosis in cervical cancer cells while being pharmacologically safe†
Abstract
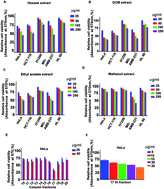
Chromolaena odorata, commonly known as Siam weed, is popular as a traditional medicine. We report the isolation and characterization of four compounds from a cytotoxic fraction, F-17, isolated from the dichloromethane (DCM) extract of C. odorata by bioactivity-guided fractionation. The organic extracts were screened in five cancer cell lines of various origins for their cytotoxic effect. The DCM extract exhibited maximum cytotoxicity and was purified by silica gel column chromatography to obtain four major compounds. The compounds were characterized by 1H-NMR, 13C-NMR, and HR-MS methods and were found to be acacetin (1), dihydrokaempferide (2), isosakuranetin (3), and kaempferide (4). MTT assay was used for preliminary evaluation of the cytotoxicity of these compounds. Among the cancer cell lines that were screened, HeLa was the most sensitive to kaempferide (IC50: 16 μM) followed by acacetin (174 μM), dihydrokaempferide (>200 μM) and isosakuranetin (>200 μM). Kaempferide (4) induced morphological characteristics of apoptosis in HeLa cells and was non-toxic to rapidly dividing normal human fibroblasts up to 100μM. Annexin V staining, characteristic of early stage of apoptosis was further confirmed by FACS analysis. Induction of apoptosis was illustrated by its potential to induce the cleavage of caspases and PARP. FACS analysis demonstrated that kaempferide (4)-induced cytotoxicity is independent of cell cycle arrest. Acute and chronic toxicity studies conducted in vivo proved that the compound is pharmacologically safe. To the best of our knowledge, this is the first study reporting the anticancer potential and pharmacological safety of kaempferide (4).

 Please wait while we load your content...
Please wait while we load your content...