Effect of annealing temperature and element composition of titanium dioxide/graphene/hemin catalysts for oxygen reduction reaction†
Abstract
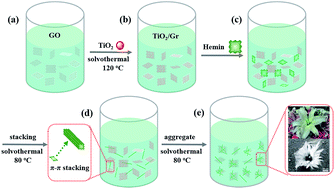
The oxygen reduction reaction (ORR) plays an important role at the cathode of fuel cells in practical applications. Herein, a titanium dioxide/graphene supported hemin (TiO2/Gr/Hem) composite material with a flower-like superstructure was successfully prepared through a two-step solvothermal reaction. By a further heat-treatment at 300–900 °C, the electrocatalytic activity of the as-obtained catalysts was examined, and it was found that the pyrolysis at 700 °C gave rise to the best catalytic activity for the ORR in alkaline media. This heat-treatment temperature was found to be crucial in determining the activity and stability of catalysts, due to the enhanced structural defects, active sites, geometrical complexity, and larger fraction of the pyridinic nitrogen and pyrrolic nitrogen groups. The titanium dioxide/graphene (TiO2/Gr) and graphene/hemin (Gr/Hem) were also studied and compared, and it was revealed that the catalytic activity of TiO2/Gr/Hem catalysts for ORR can be further enhanced. In addition, the chemically bonded element iron in the heat-treated TiO2/Gr/Hem catalysts showed an inhibition effect for ORR and Ti–C–N materials garnered high catalytic activity compared with Ti–C–N–Fe materials in alkaline media. The higher methanol tolerance and durability of the TiO2/Gr/Hem composite materials during ORR were also confirmed. These results reflected the critical influences of the pyrolysis temperature and the chemically bonded element dopants to be the key factor for ORR.

 Please wait while we load your content...
Please wait while we load your content...