Sodium alginate-based magnetic carbonaceous biosorbents for highly efficient Cr(vi) removal from water†
Abstract
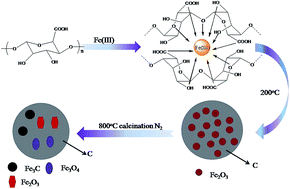
Sodium alginate-derived magnetic carbonaceous materials (Fe–SA-X, X means heating temperature) were fabricated by carbothermal reduction of ferric alginate, aiming for efficient removal of Cr(VI) from water. The materials were characterized by X-ray diffraction patterns, scanning electron microscopy, transmission electron microcopy, Fourier transform infrared spectra, X-ray photoelectron spectroscopy, nitrogen sorption, and Raman spectrometry. The adsorption conditions for Cr(VI) onto the samples were optimized, and adsorption kinetics and isotherm studies were thoroughly studied. The adsorption data fitted well with the pseudo-second-order model and Langmuir isotherm model. The Fe–SA-800 was much more efficient than both Fe–SA-400 and Fe–SA-600, and the maximum adsorption capacity was calculated to be 86.32 mg g−1 based on the Langmuir model. The adsorption mechanism was fully discussed, and desorption and regeneration experiments showed that Fe–SA-800 could be easily regenerated and reused. The above results showed that resulting magnetic biosorbents are promising for toxic Cr(VI) removal from water.

 Please wait while we load your content...
Please wait while we load your content...