Multifunctional ionic hybrid poly(propyleneimine) dendrimers surrounded by carbazole dendrons: liquid crystals, optical and electrochemical properties†
Abstract
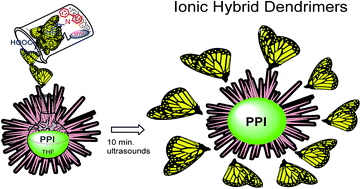
Two novel series of dendrimers synthesised by the ionic grafting of poly(propylene imine) PPI-(NH2)n (n = 4, 8, 16, 32, 64 primary amines on the surface) with bifunctional dendrons of two generations (G1COOH, G2COOH) derived from 2,2-bis(hydroxymethyl)propanoic acid (bis-MPA) were prepared. The dendrons contain a carboxyl group at their apex and electroactive carbazole groups and mesogenic units on their periphery. The occurrence of proton transfer from the carboxylic acid to the amine groups was confirmed by IR and NMR measurements. The liquid crystalline behavior has been investigated by means of differential scanning calorimetry (DSC), polarizing optical microscopy (POM) and X-ray diffractometry (XRD). All ionic dendrimers were liquid crystalline (nematic, smectic or columnar mesophases). The type of mesophase depends on the dendrimer generation as well as on the dendron generation. The electrochemical behavior of the ionic dendrimers was investigated by cyclic voltammetry (CV). Scanning electron microscopy (SEM) studies revealed that electrochemical deposition of the material on the conducting substrate takes place. The UV-vis absorption and emission properties of the ionic hybrid dendrimers have been investigated. All the compounds are luminescent at room temperature and the intensity is related to the number of carbazole units present in the molecule.

 Please wait while we load your content...
Please wait while we load your content...