Influence of pore structures of a carbon support on the surface textures of a CO oxidation catalyst
Abstract
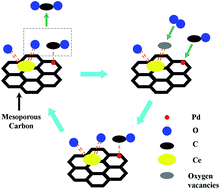
In this paper, three kinds of carbon materials, i.e., carbon spheres (non-C), microporous carbon (micro-C) and mesoporous carbon (meso-C) were synthesized. Micro-C and meso-C were prepared by adopting the microporous low silica ZSM-5 and mesoporous silica KIT-6 as templates, respectively and non-C was prepared by a hydrothermal route. The palladium loaded non-C, micro-C and meso-C supports were prepared by an impregnation method, and the catalysts were applied for CO oxidation. The Pd/meso-C exhibited the highest catalytic activity because the meso-C support had a well-ordered mesoporous structure, large BET surface area, porous channels and higher dispersion of Pd nanoparticles. By adjusting the promoter type and content, calcined temperature, the optimal prepared conditions of the catalyst were achieved: Ce loading content = 10 wt%, calcined temperature = 200 °C. Comparing the result of the characterization and activity, it was discovered that the Pd–Ce/meso-C-200 catalyst had a large number of active surface oxygen species, Ce3+ cationic species and Pd4+ cationic species. All of these were relatively conducive to the catalytic oxidation of CO.

 Please wait while we load your content...
Please wait while we load your content...