Iron encapsulated nitrogen and sulfur co-doped few layer graphene as a non-precious ORR catalyst for PEMFC application†
Abstract
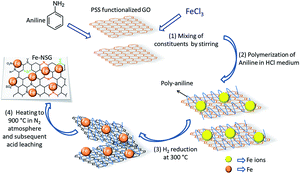
A novel strategy was followed to prepare an iron nanoparticle encapsulated nitrogen and sulfur co-doped few layer graphene (Fe-NSG) non precious electrocatalyst. For this purpose, initially graphite oxide was coated with the polyelectrolyte poly-(sodium 4-styrenesulfonate), followed by the nitrogen-containing polymer polyaniline. An iron precursor was added to this suspension and heated to 300 °C in a hydrogen atmosphere. The final heating of this nanocomposite at 900 °C in a N2 atmosphere and further acid leaching gave a non-precious Fe-NSG catalyst. X-ray photoelectron spectroscopy (XPS) data of the Fe-NSG catalyst illustrates the presence of a large amount of pyridinic and graphitic nitrogen species within the catalyst along with sulfur species. Half-cell and full cell electrochemical measurements prove the four electron transfer pathway of the oxygen reduction reaction with a high current density in an acidic environment. The special confined morphology of Fe nanoparticles within the graphene layers suppresses the agglomeration and dissolution of particles and gives long term durability. The present study illustrates a non-precious electrocatalyst for proton exchange membrane fuel cells with promising performance and stability.

 Please wait while we load your content...
Please wait while we load your content...