Highly selective CO2 adsorption performance of carbazole based microporous polymers†
Abstract
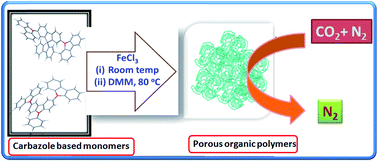
Non-coplanar shaped carbazole based monomers were used to synthesize microporous polycarbazole materials utilizing an inexpensive FeCl3 catalyzed reaction. The reactions proceed through direct oxidative coupling and extensive crosslinking polymerization routes. The obtained porous networks exhibit a maximum Brunauer–Emmett–Teller specific surface area of 946 m2 g−1 with a total pore volume of 0.941 cm3 g−1, and display a high carbon dioxide uptake capacity (up to 13.6 wt%) at 273 K and 1 atm. Selective adsorption of CO2 over N2 calculated using the ideal adsorbed solution theory (IAST) shows that these networks display enhanced selectivity with a maximum value of 155 at 298 K. Remarkably, in contrast to other materials, this value is significantly higher than the selectivity values (102–107) obtained at 273 K. Introduction of the electron rich carbazole structure into the aromatic system and pore geometry contribute to higher adsorption enthalpy which in turn leads to high selective adsorption values. These polymeric networks also show a high working capacity with reasonably high regenerability factors. The combination of a simple inexpensive synthesis approach and high selective adsorption make these materials potential candidates for CO2 storage, selective gas adsorption, and other environmental applications.

 Please wait while we load your content...
Please wait while we load your content...