Crystal engineering of ibuprofen using starch derivatives in crystallization medium to produce promising ibuprofen with improved pharmaceutical performance
Abstract
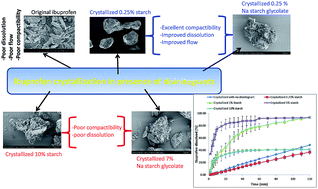
Ibuprofen exhibits poor flow, poor compaction and dissolution behaviour, and it is prone to capping after ejection from the die. Therefore, the aim of the present research was to engineer ibuprofen crystals in the presence of two disintegrants (starch and sodium starch glycolate) in order to improve its flow, compactibility and dissolution behaviour simultaneously. To this end ibuprofen and different concentrations of disintegrant (0.25 to 10% w/w in case of starch and 0.25 to 7% w/w in case of sodium starch glycolate) were dissolved in ethanol and water respectively. The ibuprofen solution was then added to the aqueous solutions containing the different concentrations of disintegrant. Ibuprofen precipitated within 10 min and the crystals were separated and dried for further studies. The obtained crystals were characterized in terms of flow, density, tablet hardness, dissolution behaviour and solid state. The results showed most of engineered ibuprofen to have better flow with a high compactibility. The results also showed that an increase in the concentration of starch in the crystallization medium resulted in a reduction in the hardness of ibuprofen tablets, but this was not the case for ibuprofen samples engineered in the presence of sodium starch glycolate. It is interesting to note that although engineered ibuprofen showed superior dissolution as compared to untreated ibuprofen, the highest concentration of starch (10%) or sodium starch glycolate (7%) slowed down the release remarkably due to an increase in the viscosity of the dissolution medium around drug particles. Solid state analysis (FT-IR, XRPD and DSC) ruled out the presence of different polymorphic forms and also any interaction between these disintegrants and ibuprofen. In conclusion, the engineering of ibuprofen in the presence of disintegrant showed how properties such as flow, compaction and dissolution behaviour can be simultaneously manipulated to suit a desired application.

 Please wait while we load your content...
Please wait while we load your content...