Impact of fluorine substitution upon the photovoltaic properties of benzothiadiazole-fluorene alternate copolymers†
Abstract
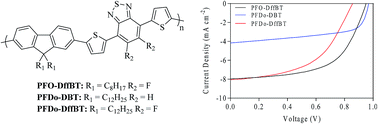
The effect of fluorine substitution on the ubiquitously-studied, moderate band gap polymer, poly(2,7-fluorene-alt-dithienylbenzothiadiazole) (PFO-DBT) has been investigated by substituting the hydrogen atoms at the 5,6-positions of benzothiadiazole to yield the polymer, PFO-DffBT. An analogous polymer, PFDo-DffBT, with longer chains on the 9H-positions of the fluorene moiety, was synthesised to rectify the low Mw of PFO-DffBT. In thin films, both fluorinated polymers display absorption bands with well-defined shoulders, giving optical band gaps of 1.91 and 1.89 eV, respectively. The optical band gap of PFDo-DBT is identical to that of PFDo-DffBT, despite the former having a significantly larger Mw. However, PFDo-DffBT displayed a blue-shifted λmax relative to its non-fluorinated counterpart. The HOMO levels of PFO-DffBT and PFDo-DffBT are lower than their non-fluorinated analogues, owing to the incorporation of electron withdrawing substituents on the benzothiadiazole moiety. The photovoltaic properties of all polymers were investigated by fabricating bulk heterojunction (BHJ) polymer solar cells using PC70BM as the electron acceptor. PFO-DffBT displayed the highest efficiency with a PCE of 4.4% despite having the lowest Mw of all polymers studied.

 Please wait while we load your content...
Please wait while we load your content...