Development of a topical adapalene-solid lipid nanoparticle loaded gel with enhanced efficacy and improved skin tolerability
Abstract
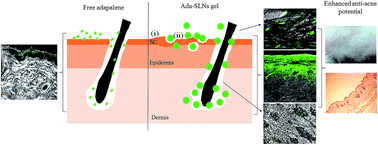
The present investigation substantiates the efficacy of adapalene loaded solid lipid nanoparticles (Ada-SLNs) in ameliorating the skin irritation potential of adapalene owing to its altered skin distribution. The Ada-SLNs were prepared by a hot homogenization technique and optimized using Box–Behnken design. Stable Ada-SLNs with a 102 ± 5 nm particle size and >85% entrapment efficiency were prepared, and thence formulated as a dispensable Cabopol gel with optimal viscosity (24.57 ± 0.27 Pa s) and high spreadability (12.39 ± 2.62 cm2). In vitro dermatokinetics revealed increased dermal bioavailability by 4.69 and 3.19 fold for 0.1% w/w Ada-SLNs gel (∼0.48 μg cm−2) and 0.1% w/w Ada-SLNs (∼0.37 μg cm−2) respectively in comparison to its free counterpart 0.1% Adiff gel (∼0.12 μg cm−2), while it was comparable (p > 0.005) to clinically superior 0.3% Adiff gel (∼0.41 μg cm−2). No detectable amount of adapalene permeated through the skin in the receptor compartment. Confocal microscopy of cryosectioned skin illustrated a significant appreciation in follicular localization of fluorophore labelled SLNs followed by its diffusion in surrounding dermis. Skin irritation studies using reconstructed human epidermis (EpiSkin) and transepidermal water loss (TEWL) revealed better skin tolerability of Ada-SLNs over Adiff gel even after higher dermal drug distribution. This visual and histological observation further clarified the enhanced anti-acne potential of Ada-SLNs gel in comparison to Adiff gel. The present SLNs can be a promising carrier for follicular delivery of adapalene along with a minimized irritation effect.

 Please wait while we load your content...
Please wait while we load your content...