Functional properties of ZnCo2O4 nano-particles obtained by thermal decomposition of a solution of binary metal nitrates
Abstract
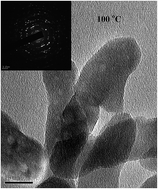
Spinel-type ZnCo2O4 nano-crystalline particles are prepared by direct thermal decomposition of a solution of binary metal nitrates at a relatively low temperatures (100 °C). Structural studies by X-ray diffraction and X-ray photoelectron spectroscopy reveal the predominant spinel crystal phase for ZnCo2O4. The high resolution transmission electron microscopy images with selected area diffraction patterns show that the spinal-type ZnCo2O4 nano-particles have a mean particle size of around 20 nm. Magnetic measurement reveals weak ferromagnetic properties for the spinel-type ZnCo2O4 nano-particles. The impedance spectroscopy measurement suggests a p-type nature for the prepared ZnCo2O4 nano-particles. The activation energies in Ar, air and O2 atmospheric conditions are found to be 0.46 eV, 0.43 eV and 0.35 eV respectively for temperatures below 200 °C. For temperatures greater than 200 °C, these samples show an intrinsic nature with activation energy of 0.57 eV for all atmospheric conditions. The ZnCo2O4 nano-crystals show a promising chemisorption process-related gas sensing behaviour for liquefied petroleum gas and they are investigated by DC and AC impedance measurements. A mechanism for the gas sensing behaviour of ZnCo2O4 is discussed.

 Please wait while we load your content...
Please wait while we load your content...