Evaluation of electrospun polymer–Fe3O4 nanocomposite mats in malachite green adsorption
Abstract
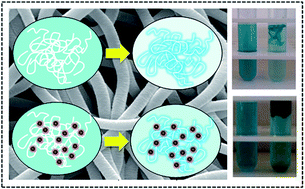
Magnetoactive nanocomposite fibers, based on poly(ethylene oxide) (PEO), poly(L-lactide) (PLLA) and pre-formed oleic acid-coated magnetite nanoparticles (OA·Fe3O4), were fabricated by electrospinning and evaluated for the first time as substrates for the adsorption of N-methylated diaminotriphenylmethane dye (malachite green oxalate, MG) from aqueous media. The adsorption of MG onto the fibers was investigated under ambient conditions by means of UV-Vis spectrophotometry as a function of initial dye concentration and solution pH. Equilibrium data for MG adsorption were well-fitted with the Langmuir isotherm model indicating a monolayer adsorption process. The effect of magnetite nanoparticles on the adsorption efficacy has been also demonstrated by performing the aforementioned studies on fibers that did not contain OA·Fe3O4. The obtained results suggested that the presence of embedded magnetite nanoparticles reduces the fiber adsorption efficiency to some extent. Moreover, the thermodynamic parameters determined from adsorption experiments carried out at three different temperatures indicated that the adsorption of MG onto the Fe3O4-free and the Fe3O4-containing fibers is spontaneous and endothermic. Although the presence of Fe3O4 within the fibers disfavored somewhat the adsorption process, nevertheless, the incorporation of the magnetic nanoparticles within these materials assisted their recovery from aqueous solutions by means of an externally applied magnetic field. Desorption of MG from the fibers could be realized upon fiber immersion in alcohol solution, thus allowing the regeneration and re-use of the adsorbents that retained the same adsorption efficiency after multiple regeneration cycles. MG adsorption studies performed in urban wastewater samples by using the PEO/PLLA and the PEO/PLLA/OA·Fe3O4 fibers as adsorbents, demonstrated the potential use of these materials in real wastewater treatment applications.

 Please wait while we load your content...
Please wait while we load your content...