One-step synthesis of three-dimensional porous ionic liquid–carbon nanotube–graphene gel and MnO2–graphene gel as freestanding electrodes for asymmetric supercapacitors†
Abstract
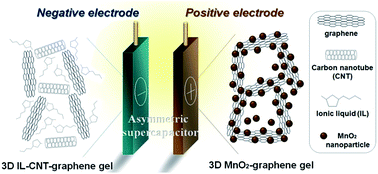
We report the design of a new type of high-performance asymmetric supercapacitor using three-dimensional (3D) porous ionic liquid (IL)–carbon nanotube (CNT)–graphene gel and MnO2–graphene gel as freestanding electrodes, both of which are synthesized by a facile and green one-step hydrothermal method. For the synthesis of IL–CNT–graphene gel, the precursor CNT–graphene oxide has firstly been dispersed by IL and then self-assembled into a unique “skeleton/skin” structure with CNTs as the skeleton and graphene nanosheets as the skin. For the synthesis of MnO2–graphene gel, the nucleation and growth of MnO2 nanoparticles as well as the self-assembly of graphene nanosheets occur simultaneously under the hydrothermal treatment, lead to a distinct “plum pudding” structure, where the MnO2 nanoparticles that are decorated on the 3D graphene gel are tightly wrapped by graphene nanosheets. In these two strategies, the introduction of IL, CNT and MnO2 nanoparticles into graphene gel not only acts as the “spacer” to prevent the π–π stacking interactions between graphene nanosheets, but also improves the electrochemical and capacitive properties of the graphene nanohybrid gel. The resultant asymmetric supercapacitor of IL–CNT–graphene gel//MnO2–graphene gel achieves high energy as well as good cycling stability and affordability, and can be reversibly charged/discharged at a maximum cell voltage of 1.8 V in 1.0 M aqueous Na2SO4 electrolyte. The corresponding energy density and power density are 25.6 W h kg−1 and 9.07 kW kg−1 at 1 A g−1, respectively, and the specific capacitance retention remains about 90% after 10 000 cycles.
- This article is part of the themed collection: Ionic Liquids: Editors collection for RSC Advances

 Please wait while we load your content...
Please wait while we load your content...