Half-sandwich complexes of ruthenium, osmium, rhodium and iridium with dl-methionine or S-methyl-l-cysteine: a solid state and solution equilibrium study†
Abstract
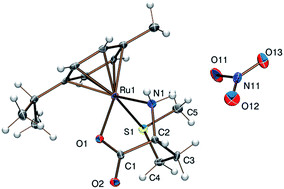
[(η6-p-cym)Ru(H2O)3]2+ binding strength of the thioether ligands DL-methionine (H2met+) or S-methyl-L-cysteine (H2mecys+) was studied with the combined use of pH-potentiometry, NMR and mass spectrometry in aqueous solution. Both ligands were found to form stable [(η6-p-cym)RuA]+ complexes with [S, NH2, COO−] coordination of the amino acids over a wide pH-range. A log βRuA value of 17.2(2) was obtained from NMR titrations for the met system by a competition reaction with hydroxide ions. Comparison of the [(η6-p-cym)Ru(H2O)3]2+ binding strength of the [O, O, O] (citrate), [O, N, O] (isoserine) and [S, N, O] (met) donor sets at pH = 7.4 reveals the exclusive formation of a [S, N, O] chelated metal complex. Synthesis and characterisation of [(η6-p-cym)Ru(L)]X, [(η6-p-cym)Os(L)]Cl or [(η5-Cp*)M′(L)]Cl (L = mecys, DL-met, L-met; X = Cl, NO3, CF3SO3; M′ = Rh, Ir) complexes together with the molecular structures of the [(η6-p-cym)Ru(mecys)]NO3 (10) and [(η6-p-cym)Ru(met)]NO3 (11) obtained by the X-ray diffraction method are reported. Both tridentate ligands form chiral-at-metal complexes in which the configuration of the metal centre is determined by the ligands containing a stereogenic centre (αC) as well. Diffraction data indicate that 10 contains the RRuRαCSS stereoisomer while 11 consists of the RRuSαCRS and SRuRαCSS enantiomers in the lattice. Owing to the labile configuration of the thioether sulfurs epimerisation in both systems was detected and the ratio of the diastereomers was estimated. 11 together with [(η6-p-cym)Ru(L-met)]Cl (3), [(η5-Cp*)Rh(DL-met)]Cl (7) and [(η5-Cp*)Ir(DL-met)]Cl (9) were tested for their in vitro cytotoxicity using human-derived ovarian cancer lines (A2780) and showed no anti-proliferative activity in the concentration range (0–50 μM) studied.

 Please wait while we load your content...
Please wait while we load your content...