Cylophosphamide elicited intracranial hemorrhage via mitochondrial ROS-hif-1α-ATP depleting pathway—preventive trials with folic acid, resveratrol and vitamin E
Abstract
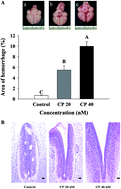
Hepatic CYP2B metabolizes cyclophosphamide (CP) into acrolein and phosphoramide mustard, which are the ultimate toxic teratogenic compounds. To determine whether folic acid (FA), resveratrol (RV) and vitamin E (vitE) could prevent these adverse effects, we carried out this study with CP at 20–60 nM. CP at 40 nM yielded maximum malformation (58.3 ± 4.2%) with a minimum mortality rate (58.3 ± 4.5%) and severe neural tube defects (NTDs). CP (40 nM) was co-administered with nutraceutics (each at 0.2 and 2.0 μM) applied to day-zero chicken embryo models (CEMs) and the biochemical parameters in brains were examined. CP upregulated (vs. control) hydrogen peroxide (HPO) (1.75/1.05 μmol per μg protein), hif-1α gene (1.5 fold), and HIF-1α protein (1.87 fold). CP downregulated glutathione (3.0/5.2 μmol per μg protein), cytochrome c oxidase (CCO) (10/22 U per mg protein) and ATP production (0.52 ± 0.7/1.23 ± 0.4 pmol per μg protein) (p < 0.05). FA and RV at 2.0 μM completely suppressed the levels of HPO to lower than 1.0 μmol per μg protein and hif-1α to <1.0 fold, but increased CCO to >20 U per mg protein, and ATP to >1.2 pmol per mg protein. As a consequence, the intracranial hemorrhage was completely alleviated. Interestingly, vitE at 2.0 μM slightly upregulated the hif-1α gene, and suppressed GSH, CCO, and ATP production, which was suggested to be due to limited permeability through the blood brain barrier and the formation of tocopheryl free radicals. Conclusively, CP tends to elicit intracranial hemorrhaging via the mitochondrial ROS-hif-1α-ATP depletion pathway. For the prevention of such teratogenicity, FA and RV at 2.0 μM are more effective than vitE.

 Please wait while we load your content...
Please wait while we load your content...