Preparation and characterization of functionalized silica spheres for removal of Cu(ii), Pb(ii), Cr(vi) and Cd(ii) from aqueous solutions
Abstract
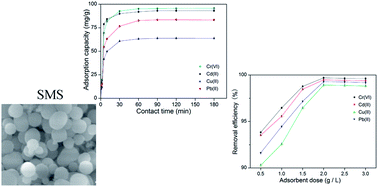
In this study, functionalized silica spheres (SMS) with a mesoporous structure were fabricated using stearic acid as a template and 3-(2-aminoethylamino)propyldimethoxymethylsilane as a cotemplate, under mild reaction conditions. SMS was prepared to develop efficient adsorbents for Cu(II), Pb(II), Cr(VI), and Cd(II) from aqueous solutions. The effect of the contact time, pH, initial metal concentration, temperature, and desorption were examined in batch experiments. The SMS displayed excellent Cr(VI) adsorption efficiency at a pH of 2. Meanwhile, the results also showed maximum adsorption of Cu(II), Pb(II), and Cd(II) at a pH of 7. Furthermore, SMS can be used repeatedly without significantly changing their adsorption capacities. In addition, the use of a stearic acid templating approach allows for the environmentally benign recovery of the cost-intensive template by simple solvent extraction. Therefore, the adsorbent SMS is expected to be an environmentally and economically feasible adsorbent for the removal of heavy metal from aqueous solutions.

 Please wait while we load your content...
Please wait while we load your content...